Phase change coolants — also known as thermal storage materials — are broadly classified into three categories based on their chemical composition: inorganic, organic, and composite. Each type offers distinct advantages and limitations in terms of latent heat, thermal conductivity, supercooling tendency, and cost.
In this first part of a two-part series, Glacier Coolant takes a closer look at inorganic phase change coolants — their strengths, weaknesses, and real-world applications.

What Are Inorganic Phase Change Coolants?
Inorganic phase change coolants include materials such as hydrated salts, ice, dry ice, liquid oxygen, and other non‑organic compounds. These materials store and release cooling energy through phase transitions — most commonly solid‑liquid or solid‑solid changes. As one of the earliest types of thermal storage media, they remain widely used today.
Ice: The Classic Choice
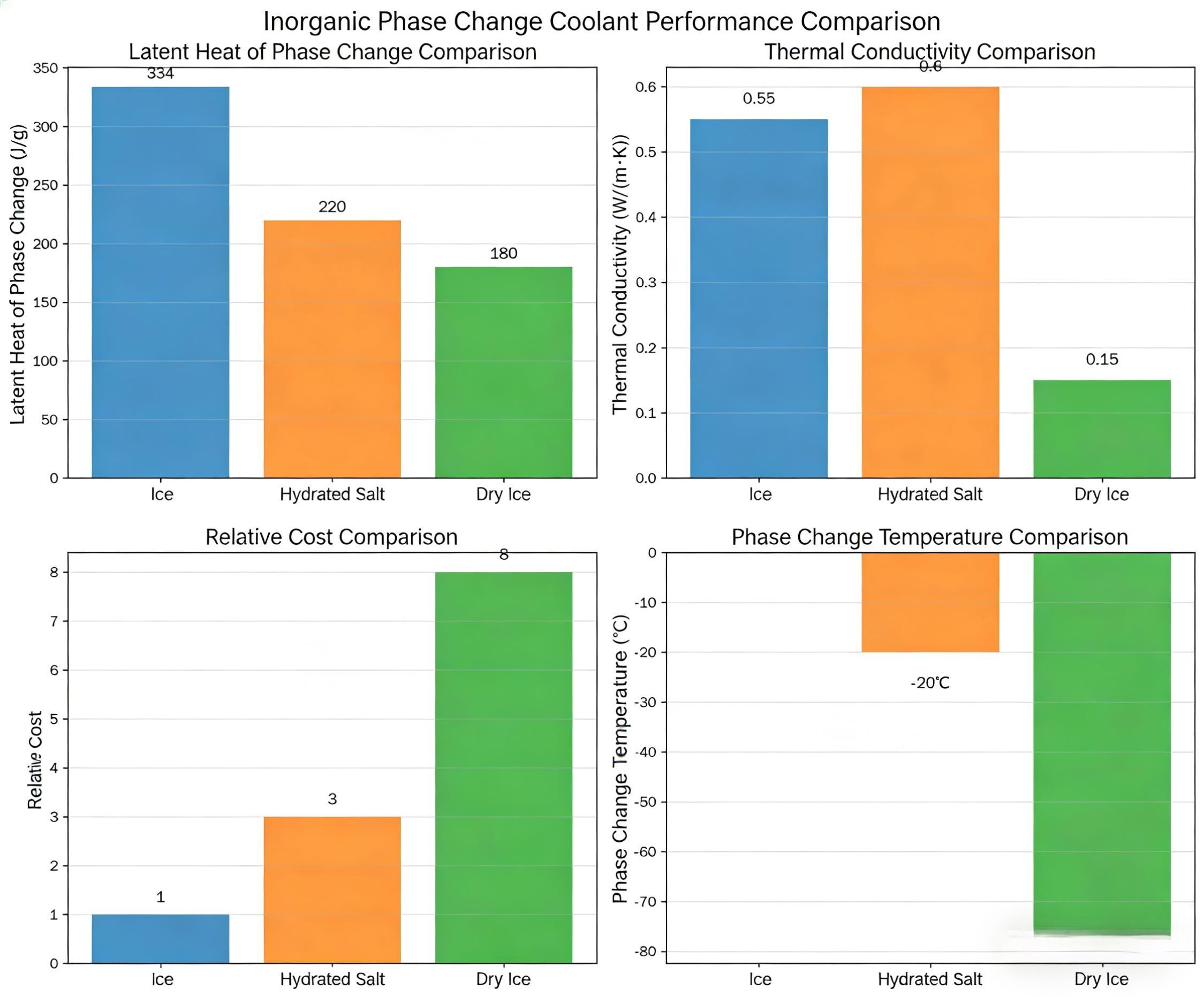
Ice remains the most familiar inorganic coolant, with a phase change temperature of 0°C.
Advantages
High latent heat: Up to 334 J/g
Widely available and naturally eco‑friendly
Extremely low cost
Limitations
Significant supercooling — requires careful control
Volume expansion during freezing can crack containers if no expansion space is reserved
Many commercial 0°C coolants are ice‑based, often improved with nucleating and thickening agents to reduce supercooling and manage volume changes.
Hydrated Salts: The Market Leader
Hydrated salt coolants are currently the most commonly used phase change materials in the market.
Advantages
High latent heat – typically exceeding 200 J/g
Good thermal conductivity
High cooling density per unit volume
Low cost and widely available raw materials
Safe and low toxicity
Adjustable phase change temperature – can be tuned from –80°C to 0°C by varying the salt type and concentration, making them adaptable to many applications
Limitations
Significant supercooling – requires nucleating agents to control
Phase separation after repeated cycles – anti‑separation additives are needed
Corrosion potential – long‑term use can corrode metals; protective coatings add cost
Glacier Coolant note: With proper formulation and system design, many of these issues can be effectively managed.
Dry Ice: The Ultra‑Low Workhorse
For ultra‑low temperature cooling (typically –80°C to –50°C), dry ice is the most widely used solution.
Phase change temperature: –78.5°C
Mechanism: Sublimates directly from solid to gas, absorbing heat
Advantages
High cooling density
Non‑toxic and non‑polluting
No leakage risk (solid form)
Chemically stable
Limitations
Relatively high cost
Requires careful handling (frostbite risk, ventilation needs)
Liquid Oxygen: For Cryogenic Applications
When temperatures drop below –100°C, liquid oxygen becomes the coolant of choice in cryogenic applications.
Mechanism: Vaporization of liquid oxygen absorbs heat to maintain ultra‑low temperatures
Advantages
Widely available
Stable performance
Environmentally friendly
Limitations
Difficult and costly to transport
Requires specialized handling and storage equipment
Summary Comparison
| Coolant Type | Phase Change Temp. | Key Strength | Main Challenge |
|---|---|---|---|
| Ice | 0°C | High latent heat, low cost | Volume expansion, supercooling |
| Hydrated salts | –80°C to 0°C | Adjustable temp, high density | Phase separation, corrosion |
| Dry ice | –78.5°C | High density, no leak risk | Cost, handling safety |
| Liquid oxygen | Below –100°C | Stable, eco‑friendly | Transport difficulty |
Glacier Coolant: Your Partner in Thermal Storage
With deep expertise in heat transfer and phase change technologies, Glacier Coolant helps customers select or formulate the right coolant for their specific needs — whether it's ice‑based, hydrated salt, or advanced organic/composite blends.
In the next installment, Glacier Coolant will explore the advantages and limitations of organic and composite phase change coolants.
