Glacier Coolant Clarifies the Trio Behind Every Cooling System
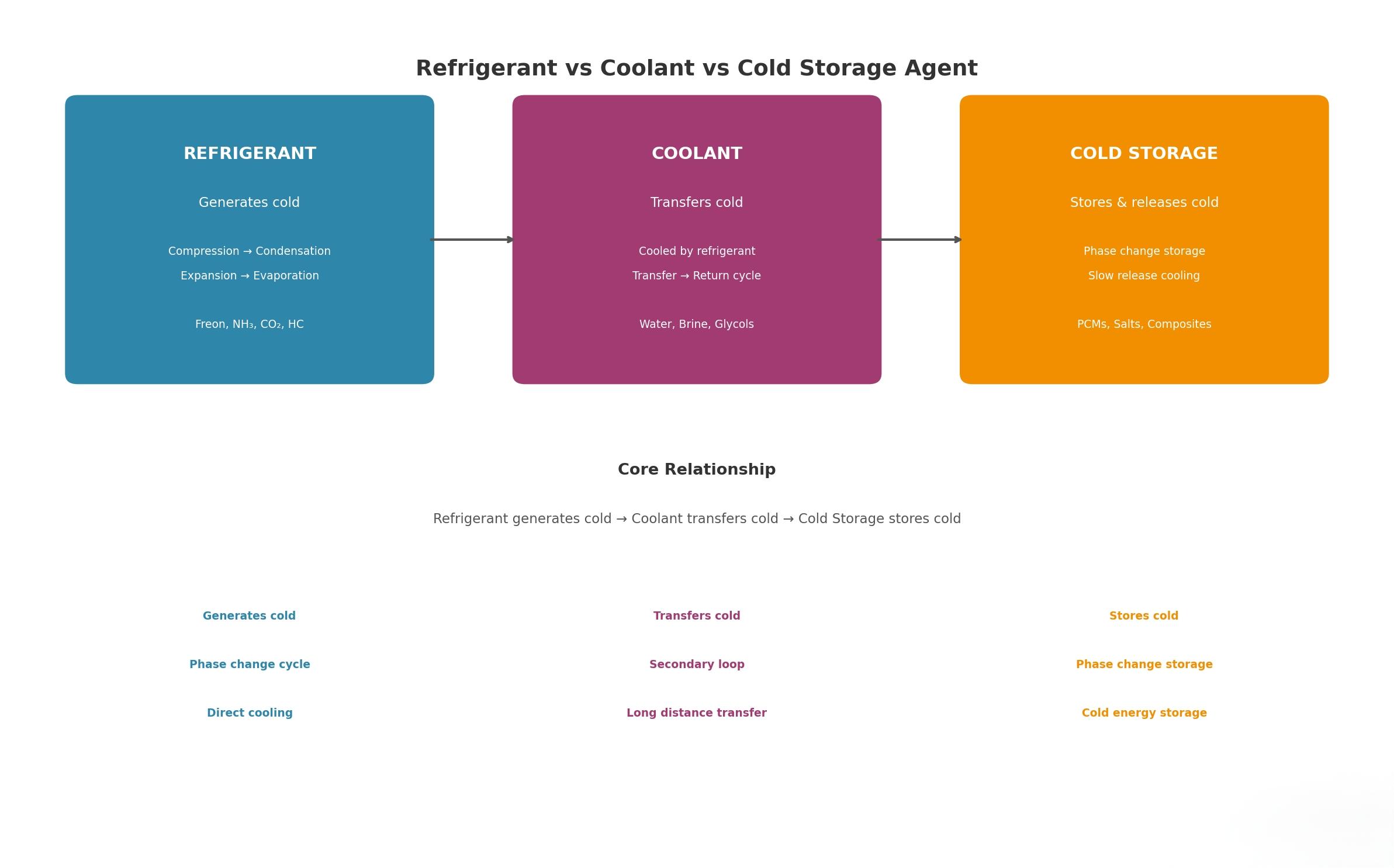
Behind every effective cooling system — whether in a supermarket freezer, a pharmaceutical cold chain, or an industrial reactor — lie three very different but closely related substances: refrigerants, heat transfer fluids (often called secondary coolants), and phase change materials (PCM). Glacier Coolant breaks down what each one does and how they work together.
Refrigerants: The Workhorses That Create the Cold
Refrigerants are the substances that actually generate cooling. They undergo a continuous cycle of compression, condensation, expansion, and evaporation — changing from gas to liquid and back again. Each phase change absorbs or releases heat, producing the cold effect that makes refrigeration possible.
Common examples include ammonia, carbon dioxide, hydrocarbons, and various fluorinated gases.
Without a refrigerant, there is no cooling. It is the engine of any mechanical refrigeration system.
Heat Transfer Fluids: The Messengers That Move the Cold
While the refrigerant generates cold inside the evaporator, it is often not practical to circulate refrigerant directly to distant or multiple cooling points. That is where heat transfer fluids — also called secondary coolants — come in.
These fluids are first chilled by the refrigerant inside the evaporator. They then travel through pipes to the equipment, product, or space that needs cooling. After absorbing heat from the target area, they return to the evaporator to be cooled again.
Water, brine, ethylene glycol, and propylene glycol are typical examples. Their job is purely transport — they carry cold from point A to point B.
Phase Change Materials (PCM): The Reservoirs That Hold the Cold
Phase change materials play a different role: storage. Instead of continuously circulating, PCM absorb and store cold energy when temperatures are low, then release that stored cold when temperatures rise — often without any active pumping or external energy.
This makes PCM ideal for applications where temperature must remain stable over time, such as vaccine transport, food cold chains, and thermal storage air conditioning systems.
PCM can be organic (paraffins, fatty acids), inorganic (salt hydrates), or composite blends that combine the advantages of both.
Interestingly, some heat transfer fluids can also serve as PCM, and some PCM can be formulated to flow like fluids — blurring the lines between the two categories.
How They Fit Together
| Substance | Primary Job | Moves? | Stores Cold? |
Refrigerant | Generates cold | Yes (circulates within sealed loop) | No |
Heat Transfer Fluid | Transports cold | Yes (pumped to load) | Sometimes |
PCM | Stores cold | No (fixed or encapsulated) | Yes |
In a typical industrial cooling system:
The refrigerant creates the cold inside the chiller or evaporator.
A heat transfer fluid carries that cold to multiple points of use.
PCM can be placed at those points to maintain stable temperatures during interruptions or peak demand.
All three are complementary — not competing. A well-designed system may use two or even all three together to achieve precision, efficiency, and reliability.
Glacier Coolant: Your Partner Across the Cooling Spectrum
Glacier Coolant offers a broad portfolio of heat transfer fluids and phase change materials for applications ranging from –110°C to 340°C. Whether you need a low-conductivity fluid for sensitive electronics, a non-toxic coolant for food processing, or a reliable PCM for cold chain logistics, Glacier Coolant provides solutions backed by decades of experience and ISO-certified quality.
Understanding the difference between refrigerants, heat transfer fluids, and PCMs is not just technical knowledge — it is the key to designing smarter, safer, and more energy-efficient cooling systems.
