Water serves a versatile function in refrigeration and cooling processes, acting either as a coolant or as a refrigerant depending on the system design and operating conditions.
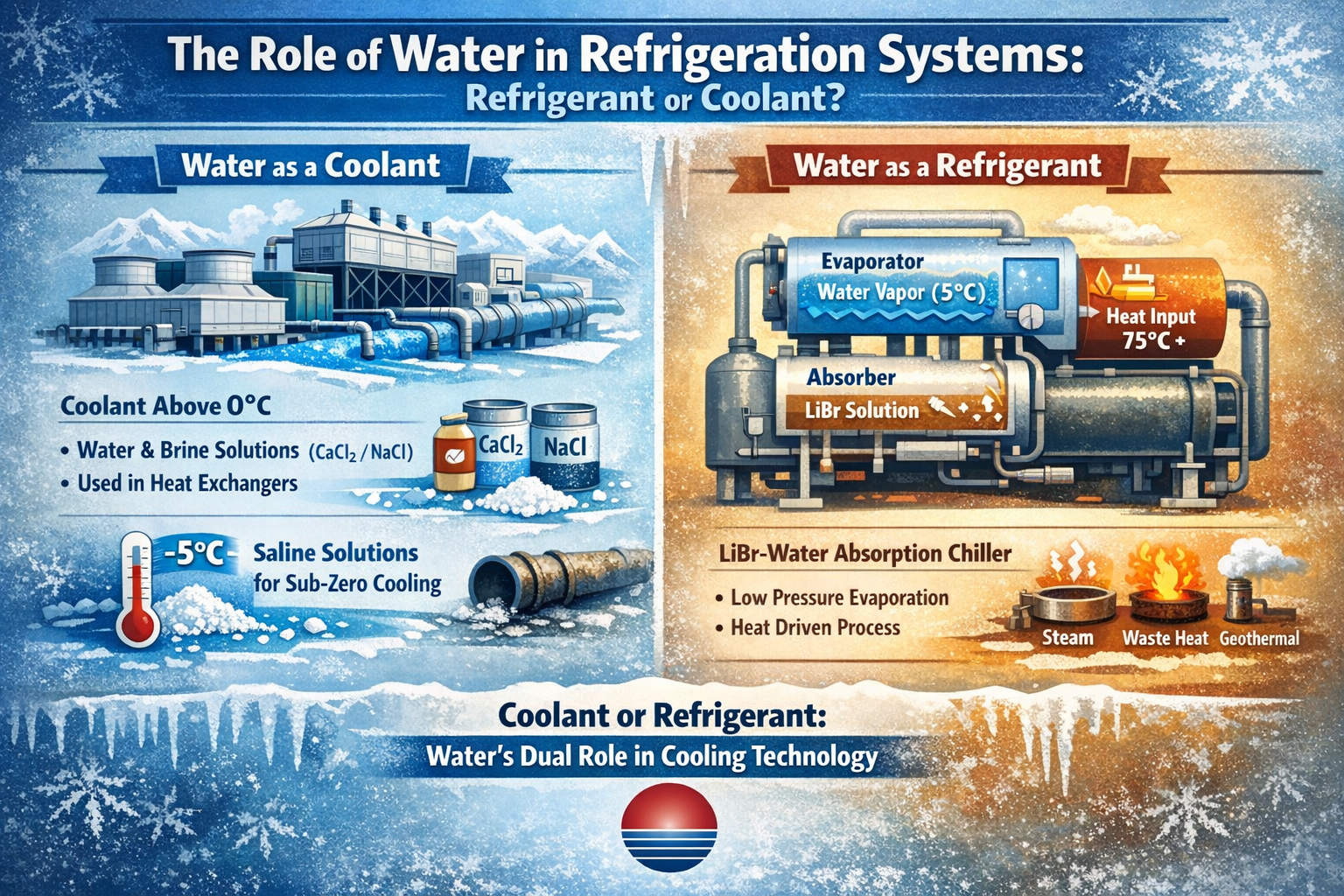
Water as a Coolant
Water is commonly employed as a coolant in applications where the operating temperature remains above 0°C. It offers several advantages, including a high specific heat capacity, efficient convective heat transfer, and low cost. For sub-zero temperature operations, saline solutions—typically prepared by dissolving calcium chloride (CaCl₂) or sodium chloride (NaCl) in water—are utilized as coolants. As the concentration of salt increases, so does the density and flow resistance of the solution, while its specific heat decreases. This requires a larger flow rate of the saline solution to transfer the same amount of cooling energy, consequently increasing pump power consumption. Therefore, when preparing saline solutions, it is essential to ensure that the freezing point of the mixture is slightly below the lowest expected system temperature—generally 5–8°C lower than the refrigerant's evaporation temperature. It is also important to note that saline solutions can be corrosive to metals, particularly when slightly acidic and exposed to air.
Historically, water and brine were the primary coolants in many industrial applications. However, modern alternatives such as Glacier Coolant have been developed, offering improved characteristics like higher specific heat, lower viscosity, and greater stability.
Water as a Refrigerant
Water can also function as a refrigerant, notably in absorption refrigeration systems such as those using lithium bromide (LiBr)-water as the working fluid pair. Similar to vapor-compression refrigeration, absorption systems rely on the evaporation of a liquid refrigerant at low pressure and temperature to absorb heat from a coolant (typically chilled water). The key distinction lies in the use of a binary working fluid—here, the LiBr-water solution—where water acts as the refrigerant.
In a lithium bromide absorption chiller, water serves as the refrigerant, evaporating under vacuum conditions (absolute pressure around 870 Pa) at a low temperature (approximately 5°C). This process absorbs heat from the coolant, thereby producing chilled water continuously. Lithium bromide, the absorbent in the mixture, has a strong affinity for water vapor at low temperatures but releases moisture when heated. This cyclical absorption and desorption of water refrigerant enables continuous cooling operation. The necessary thermal energy for regeneration can be supplied by steam, waste heat, exhaust gases, geothermal hot water (above 75°C), or direct combustion in fuel-rich settings. These features contribute to the economic appeal and growing adoption of LiBr absorption chillers.
From a thermodynamic perspective, any liquid undergoing phase change from liquid to vapor absorbs heat from its surroundings—known as the latent heat of vaporization. The boiling temperature of water decreases with reduced pressure; for instance, at atmospheric pressure it boils at 100°C, while at 0.05 atm it boils around 33°C. By creating a low-pressure environment where water can evaporate and absorb heat, it becomes feasible to use water as an effective refrigerant in suitable applications.
Glacier Coolant specializes in the research and application of both coolants and refrigerants for advanced thermal management solutions.
